Particle-Size-Analysis of fine-comminuted tin ore
My big announcement!
First particle-size-analysis on an ore I have comminuted in my rod-mill!
I will return to explaining the meaning(s). Before that...
What this looks like:
sieved to size fractions in bowls to front; decanted very fines to
rear

Getting there...
To now...
-
I made the
rod-mill
[my YouTube video - link current 17Mar2026], ["911metallurgist" links:
*,
*]
which ground (comminuted) the tin ore
-
I comminuted some galena ore, leading to metallic
lead
in-hand - but no particle-size-analysis...
-
main training was comminuting "builders merchant" granite chippings
then particle-size-analysis of what resulted - webpages
*,
*
-
"granite chippings" programme introduced sieve-analysis [911metallurgist:
*
] for particle-size-analysis - "the workhorse of mineral processing" -
and challenges had me learn
sedimentation with decantation
(has external links to topic)
-
trying to separate the cassiterite (tin mineral) from fine ore on a
shaking-table [external:
Wikipedia,
Holman Wilfley]
had me see very-fines ("slimes") must be separated/removed in a
pretreatment of the ore - which with sedimenation separation
brought in the need for deflocculants - encountered
"washing-soda" (sodium carbonate) and SHMP (sodium hexametaphosphate)
Specific to the ore featuring here...
-
assertion "finer is better", which I duly fulfilled at that time, is
not generally held to be the case...
-
there's two samples of the same comminute here - the "650g" and the
"200g" - that the results are the same is good and implies my sampling
is good and honest
-
ore pre-treatment: seeking deflocculation assisting "desliming"
- "650g" sample was treated with Na2CO3 - then "decanted" in a full
trug - which should have ensured "Stokes settling"
- "200g" sample was treated with SHMP - then decanted in a tall
narrow 1.5L container - giving a turbid supernatant with fines
concentration in water - was "Stokes settling" or "hindered" to
some extent?
-
the as-comminuted ore "smokes" when spooned around (not a good sign
for several reasons - mineral-processing and environment) [the
decanted very-fines dry cannot be handled in any enclosed space - even
slowly dragging a teaspoon through it side-on, acting like a
plow-share, raises a lot of dust - reason all industrial handling is
in water as a slurry]
"Stats" about the ore
- there was 4.5kg of the ore; hence 4.5kg of comminute
- it is seen to have tin (cassiterite) in it - that reveals
sometimes when handling in water - eg. falling drip-by-drip into a
trug
- the tin mineral is hosted in quartz - there is black substance
within, postulated to be tourmaline - so a typical Cornish ore
Specifics
Do "deslime"
I was advised:
-
"desliming" would definitely lose the very very fine cassiterite
to the "slimes" (though might be unrecoverable anyway)
-
"slimes" (very fines) coagulating on all particles including
cassiterite particles may "blunt" gravity-separation (best separation
method currently for tin), reducing recovery - which likely would
cause more cassiterite loss to tailings than if some loss in
"desliming" were accepted
-
the "slimes" muddying the slurry/water flowing across the
shaking-table make it difficult to see what is happening - which I
observed doing a trial run - and for a novice/learner I needed to make
the task more obvious and achievable
Another very pragmatic reason to "deslime"...
If you are going to sieve, performing sieve-analysis as
particle-size-analysis, you must remove the fines else you will be
choking on dust as you try to sieve (if done dry) - and that dust
contains a predominantly silica, all recently fractured (not weathered
at all - if that makes a difference?) - which is a severe hazard.
Have "slimes" - very fines - in the comminute and that becomes far
further worse...
So: "deslime".
"Decantation" to "deslime"
What I do is a scaled-up version of "beaker decantion", and it works.
It looks crude; uses much increasing more water as your "cut" gets
finer (you cannot re-use the water quickly, as the very-fines don't
sediment out quickly enough in a day and quick re-use would
re-introduce what you most want to remove); is time-consuming; and you
get wet(!).
But yes it works and tentative indications are that it is accurate (in
the "granite" tests where the Stokes' calculation said 10 seconds wait
and did give 100% sieve pass of opaque turbidity supernatant, a 6
second wait resulted in some above-75micron on the sieve - whether
that accuracy is maintained at smaller sizes is unverified).
As mentioned - specifying the outcome of decantation comes from
applying
Stokes calculations
My "cut" is at a Stokes sedimentation velocity of 1.3mm/s.
Being in water, this should give a "cut" at
- 38microns "sphere-equivalent" for silica/quartz
- 20microns "sphere-equivalent" for cassiterite
given their different densities.
The ratio of sizes at the "cut" is to-advantage
- only finer cassiterite is separated as slimes
- at a tolerable cassiterite loss, a bigger fraction of silica is
removed
Deflocculation
Taking this on-trust at the moment - "control tests" I could do to
observe the effects are yet to be done.
I have observed "hindered settling" - it is vastly slower than "Stokes
settling" - in the supernatant after vigorously stirring ore-in-water
("slurry") when the deflocculants tried were present - which I have
not observed before. Promising... Comparison with a control of
"untreated" comminute is one "control".
The amounts needed per small masses of ore are so small that I made-up
"stock-solutions" and used a small syringe-barrel to dispense
millilitre dosant quatities.
What is displayed, and what is seen
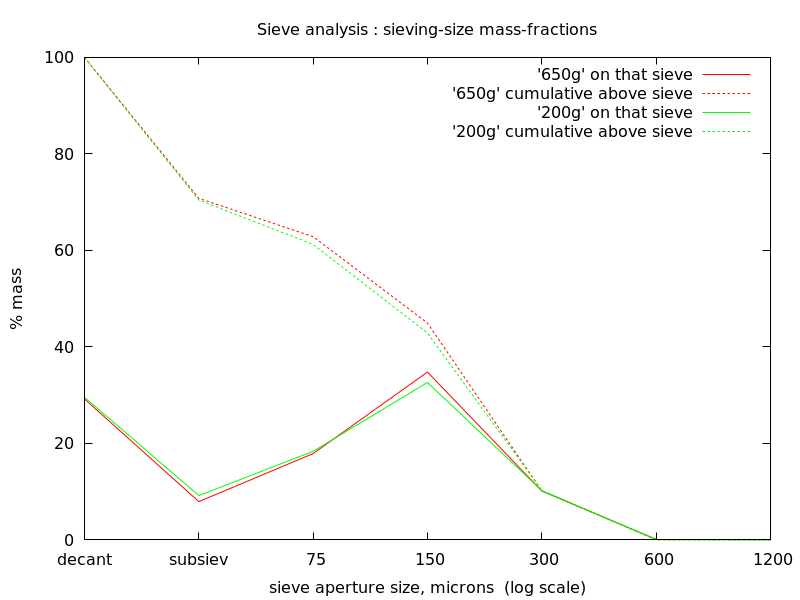
Redisplaying the plot:

A caution - my weighing-machine is accurate to only 1g - not fine
enough for any differences to be seen to have significance.
These are rough tests using crude means; there is no case to use
sophisticated expensive equipment - and these tests have got done...
Observations
- the horizontal scale here has a range covering mineral separation
sizes
- "sub-sieve" (subsiev) is plotted semi-arbitrarily at 37.5microns;
likewise "decant" (decanted) is plotted at 18.75microns
- as previously noted - that the "650g" and "200g" samples graph
very-closely the same shows:
- my sampling is honest and true
- if there is difference in the efficacy of the two
deflocculants, SHMP and Na2CO3, it is not revealed in masses at
separation sizes (probably nor would it be expected to be - very
troublesome very-fines may not amount to an observable percentage
change in separated masses at sizes - the effect would be most
expected at the "sub-sieve" size
- 150microns particle-size has the largest mass-fraction
- there is a large mass-fraction in the very-fines removed during
pretreatment by decantation - which in any commercial sense would
indicate that control of comminution needs to be improved
Detailed dicussion(s)
Validity of "sub-sieve" and "decanted" positions on the horizontal
axis:
Stokes calculations specify the decantation to "cut" at 38microns
quartz - so the decanted supernatant *should(!)* 100% pass a 38micron
sieve had one been available.
Hence the solids in the supernatant decanted are put at the next sieve
size down in the "halving" series.
The justification begs proof that assumptions are holding-true.
Overall...
At the time of writing (afternoon of Tuesday 17 March 2026) I am
pleased. I am obtaining data by sieve-analysis, processing it, and
graphing the data using "scripts" in a "unix toolkit"
working-environment on a computer.
Which hopefully means that new data can be processed through the same
"pipeline" (that is a recognised term working at a "unix" command-line
environment).
On the computation side - with the data being text-table files, and
the processing being via "scripts" picked-up by the familiar
"unix-environment" programs, the files to store are very small and
easy to retain. Also the "unix toolkit" programs have remained
unaltered at the user-interface in time over decades, so compatibility
in time has never proved a problem.
(R. Smith, 16Mar2026 to 17Mar2026, 22Mar2026 (pic.siev,decant))



